232 Courses
-
Philosophy in Film and Other Media
-
Philosophy of Love in the Western World
-
Power and Negotiation
-
Practical Information Technology Management
-
Practice of Finance: Advanced Corporate Risk Management
-
Principles and Practice of Drug Development
-
Principles and Practice of Tissue Engineering
-
Principles of Chemical Science, Advanced Track
-

Atomic Theory of Matter (Part 2)
-

Discovery of Nucleus (Part 2)
-
Wave-Particle Duality of Radiation and Matter
-
Particle-Like Nature of Light (Part 2)
-
Matter as a Waves
-

Schrödinger Equation for H Atom
-

P Orbitals (Part 2)
-

Hydrogen Atom Wavefunctions (Part 2)
-
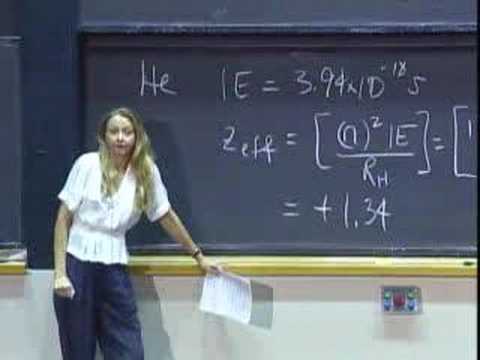
Electronic Structure of Multielectron Atoms (Part 2)
-

Periodic Trends in Elemental Properties (Part 2)
-

Why Wavefunctions are Important?
-
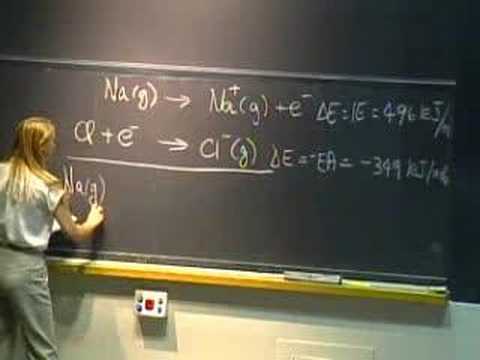
Ionic Bonds - Classical Model and Mechanism
-

Kinetic Theory - Behavior of Gases
-

Distribution Molecular Energies
-

Internal Degrees of Freedom
-
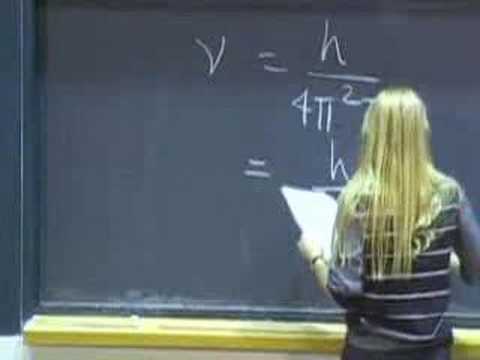
Intermolecular Interactions
-

Polarizability
-

Thermodynamics and Spontaneous Change
-

Molecular Description of Acids and Bases
-

Lewis and Bronsted Acid-Base Concepts
-

Titration Curves and pH Indicators
-

Electrons in Chemistry: Redox Processes
-
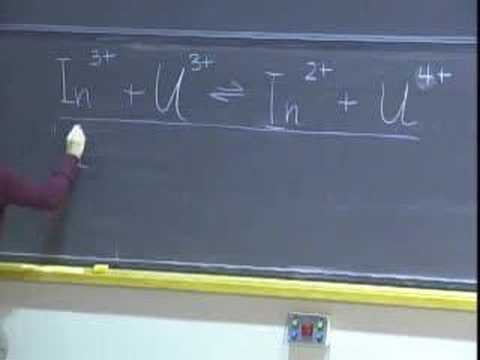
Cell Potentials and Free Energy
-

Theory of Molecular Shapes
-

Valence Bond Theory
-

Molecular Orbital Theory (Part 2)
-

Molecular Orbital Theory for Diatomic Molecules
-

Molecular Orbital Theory for Polyatomic Molecules
-

Crystal Field Theory (Part 1)
-

Crystal Field Theory (Part 2)
-

Color and Magnetism of Coordination Complexes
-
Coordination Complexes and Ligands
-
Ligand Substitution Reactions: Kinetics
-

Bonding in Metals and Semiconductors
-

Nuclear Chemistry and the Cardiolite Story
-
-
Principles of Chemical Science, Normal Track
-

Atomic Theory of Matter (Part 1)
-

Discovery of Nucleus (Part 1)
-

Wavelike Properties of Radiation
-
Particle-like Nature of Light (Part 1)
-
Matter As a Wave
-

The Hydrogen Atom
-

Hydrogen Atom Wavefunctions (Part 1)
-
P Orbitals (Part 1)
-
Electronic Structure of Multielectron Atoms (Part 1)
-

Periodic Trends in Elemental Properties (Part 1)
-

Covalent Bonds
-

Lewis Diagrams
-

Breakdown of Octet Rule
-
Molecular Orbital Theory (Part 1)
-

Valence Bond Theory and Hybridization
-
Hybridization and Chemical Bonding
-

Bond Energies / Bond Enthalpies
-

Free Energy of Formation
-
Chemical Equilibrium
-

Chemical Equilibrium (cont.)
-
Acid-Base Equilibrium
-

Acid-Base Equilibrium (cont.)
-

Acid-Base Equilibrium: Titrations
-

Acid Base Titrations and Oxidation/Reduction
-
Oxidation/Reduction
-

Oxidation/Reduction (cont.)
-

Transition Metals 1
-
Transition Metals 2: Crystal Field Theory
-

The Shapes of Molecules: VSEPR Theory
-

Kinetics 1
-

Kinetics 2
-

Kinetics 3
-

Kinetics 4
-
Kinetics 5: Catalysis
-

Review for Principles of Chemical Science, Normal Track
-
Transition Metals 3
-